Andrew Cutler and Professor Brian Mazzeo, Electrical Engineering
Purpose:
To build a robust planar device that can take an in situ, non-destructive conductivity reading on thin cathode films.
Introduction:
Li-ion batteries are an omnipresent part of modern life. Because of their high energy density and performance after repeated cycles they have become the battery of choice for portable electronics as well as electric vehicles. However, even after over 30 years of research and production of Li-ion batteries, there are still some mysteries about their functionality. One shortcoming of the current production of batteries is not knowing the conductivity of a cathode until production is complete. Even after production the quality check for cathode conductivity is generally indirect, removing a sample of batteries from a given batch and testing how they perform. If the batteries are sub-par, it can be inferred the electrode is inferior. A fast, nondestructive method to test cathode performance could overcome this.

The four point probe testing method, which puts a current through two outer leads and measures a voltage between the center two, has been around for 150 yrs. Using the measured current and voltage, along with some geometry, a conductivity can be found. It’s useful in that it allows one to ignore contact resistances, which, especially for very small geometries, may be a substantial portion of the measured resistance. However, the spacing of the probes is an issue when working with thin films on a current collector. If the outer two probes are not closer together than the height of the film, the current will take the path of least resistance through the film, which tends to be across the current collector. Thus micro four point probes have been explored. Other efforts have met considerable difficulty. Four exact points on that scale are so fragile other groups have had to take their measurements in a vacuum chamber, significantly complicating the procedure. Answering these concerns, we have built a probe with four lines instead of points. (See figure 2). This allows a much sturdier planar device, while still preserving the basic principles of a four point probe.

Methodology:
The probe is fabricated in the clean room. Using a standard liftoff process four nickel lines, each 10um wide and separated by 10um, are etched on top of a glass wafer. Then the entire wafer but a window of 70 by 500 microns is covered in a protective layer of a cross-linked polymer SU-8. To bring the four lines above the SU-8 layer they are electroplated with a layer of copper followed by gold.
The probe is mounted on a computerized, movable XYZ stage. The electrode sample is placed on a pressure sensor below. This setup allows the sample to be pressed against the probe, data gathered, then removed from contact. The pressure sensor ensures safety for the device and sample. Our protocol has been to take twenty repeat measurements in each place to verify that the data is consistent. After each set of measurements the stage moves the probe to different location. Stitching these points together we can make a map of the conductivity.
Results:
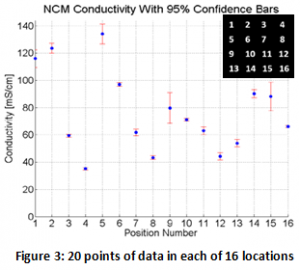
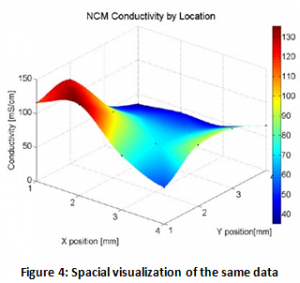
Battery electrodes have more variability than previously expected. Figures 3 and 4 show the spatial variability in conductivity on a commercial Nickle Cobalt Manganese (NCM) cathode. Each point in figure three represents twenty points of data with confidence bars. Notice that on most of the points the confidence bars are small, or, in other words, data taken from the same spot is essentially the same at each contact with the probe. Figure 4 visualizes the data spacially making it clear that there the points of high conductivity are clustered together in a “hot spot” where most of the current flows. These hotspots could cause heating and lead to failure of the battery cell.
Discussion:
This data has application in industry. Current design of electrodes (see Figure 1) uses bulk, or average, conductivity. Our data showing the variability implies that this is not a useful assumption and ignoring it requires over-engineering of batteries so that they can perform at the same level. Taking measures to decrease the variability, such as using smaller particles or whatever else future researchers can discover, could solve those problems.
Conclusion:
The micro four-line probe can take non-destructive conductivity measurements on thin film battery electrodes. This has applications in our understanding, modeling and design of future batteries.
