Tara Pandey and Dr. William Pitt, Department of Chemical Engineering
Cancer is among the leading causes of death in the world. The goal for this project was to visualize a liposome containing perfluorocarbon emulsion an eLiposome, inside of tumor cells using confocal and fluorescence microscopes. A successful study of this project would help to understand the behavior of cancer cells in the presence of drugs during targeted drug delivery for cancer.
Experiment, Results and Future Work
The project utilized lab instruments both from the department of chemical engineering and the life science for preparation and characterization of eliposomes. Confocal microscope was used for quality analysis and quantification of drug inside cells. Fluorescence microscope was used for tracking the drug inside the eLiposomes in HeLa cells. A successful study of drug inside the cells involved many complex steps and required detailed understanding of biological phenomena of cells. The project used the expertise from Jon Hartley in modifying the eLiposomes by attaching polyethylene glycol(PEG) to avoid elimination of drug carriers from reticuloendothelial system. However, due to limited time, the project could not solved many problems that arose during the cellular uptakes of drug carriers inside of cells.
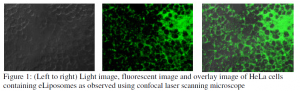
Experiments were conducted using calcein (green) and fluorescein dextran to track drug inside eLiposomes and endosomes inside the HeLa cells, respectively.
Confocal microscope image analysis shows very small number of cells (ca. 20%) with green calcein; mostly around the cell membranes. The figure 1 above shows that most of the fluorescence is also localized outside of the cells. Lower drug uptake by eLiposomes was a big problem faced for the study of the project. Before further work have been done to study the effect of ultrasound in targeted drug release in cancer cells, a higher eLiposomal endocytosis was required. For the higher uptakes, a further work could be performed to modify the surface of eLiposomes in order to achieve the desired endocytosis. Modification of the surface by attaching PEG on the surface has shown to increase the endocytosis [1]. Excess outside calcein and fluorescence was seen outside the cells (shown in Figure 1 above). This means minimum uptakes and poor cleaning of outside calcein from outside eLiposomes. The outside calcein was removed using centrifugation technique. The process was fast and effective but the dynamic light scattering showed that the use of centrifugation leads eLiposomes to coalesce together making bigger sized eLiposomes. The bigger size means a significantly reduced cellular uptakes. Hence, yet another area that need to be focused is the size of drug carrier controllability because smaller sized eLiposomes facilitate the endocytosis. Other methods of calcein removing like dialysis (using membrane to separate calcein from eLiposomes) and spin column could be other options to see if calcein could be separated without increasing the size of eLiposomes. However, these techniques were not carried out for the project.
HeLa cells were incubated with eLiposomes for 24 hours. Studies could be done with different time duration to see the effect of time in endocytosis. Ultrasound (20kHz frequency) was used to burst open drug carriers inside the cells. The effect of US was analyzed using the flow cytometer data (not shown here) analysis. A very little change in green color after US showed a little or no effect of ultrasound. This could be because a very small numbers of cells had eLiposomes with calcein inside them. This is why much of work was done in trying to achieve successful endocytosis of drug carriers inside HeLa cells. The experience gained and some preliminary observation seen are valuable for further studies. This has helped understand some fundamental issues to worry about when performing the similar experiments again.
Conclusion
The ORCA grant for this project provided an opportunity to understand the challenges that need to overcome for successful targeted drug delivery for cancer. Most of the time was spent in studying the cellular uptakes of drug carriers using confocal microscope. Even though satisfactory cellular uptake results were not observed during the experiments, our team were benefited with the project in comprehending and identifying areas needed to focus for further studies. The poor endocytosis of eLiposomes by HeLa cells was the major problem observed and this could be solved if further studies is carried out paying attending to better technique to wash external calcein from eLiposomes and finding a better method to bind eLiposomes to cell membrane to avoid any possible elimination of cells by RES. We have prepared a lab manual (available in Dr. William Pitt’s lab) with all the procedure and steps involved in this study and we hope this will facilitate a new researcher in this area.
Acknowledgements
I am very grateful to the ORCA grant for providing such an opportunity to work in this project. I wish to thank Dr. William G. Pitt for guiding me throughout the project and Jon Hartley for his help in performing many of the experiments.