Adam M. Bowman and Dr. Matt A. Peterson, Chemistry and Biochemistry
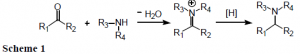
Reductive amination is an important method used to generate amines from aldehydes or ketones. This process occurs in a two-step in situ reaction. First, an iminium intermediate is formed when the carbonyl compound condenses with ammonia or a primary or secondary amine. This intermediate is then reduced by a reducing agent already present in the reaction mixture (Scheme 1).
Several reducing agents have been utilized to carry out reductive amination reactions. Some of these include: pyridine-borane, sodium cyanoborohydride, sodium triacetoxy borohydride, zinc borohydride-zinc chloride, and hydrogen in the presence of transition metal catalysts. These reagents have proven successful for a range of substrates, but each possesses limitations. For example, sodium cyanoborohydride produces toxic byproducts after aqueous workup. Hydrogenation reduces other functional groups including nitro, cyano, or carbon-carbon multiple bonds. Zinc borohydride-zinc chloride and sodium triacetoxy borohydride are ineffective with hindered alkyl or aryl ketones, and pyridineborane is only effective under acidic conditions and also requires fairly harsh workup conditions.
While pursuing research involving the synthesis and evaluation of reagents for asymmetric reductive amination, we discovered that benzylamine-borane is an effective reducing agent for this reaction. Because of the limitations associated with other reductive amination methods, as well as the mild conditions employed with benzylamine-borane, this new protocol may prove to be the method of choice in many circumstances.
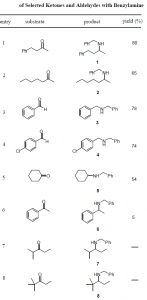
Benzylamine-borane is an air stable complex that may be stored at room temperature without observable loss in reactivity for up to one week. It is prepared by adding one molar equivalent of BH3-THF to benzylamine in THF and may be used in either protic or aprotic solvents. It efficiently reduces aldehydes and ketones (t1/2 = 3h for reduction of 0.2 M benzylacetone in THF), but in the presence of one equivalent of benzylamine and activated 4 angstrom molecular sieves it promotes smooth reductive amination to give secondary amines in good yields (Table 1).