Joseph Johnson, Department of Chemistry and Biochemistry
Introduction
Although iron Is necessary for life, It Is toxic If left free In most organIsms. To control Its toxic effects and still have iron available for biological use, organisms use ferrltins. Ferritins are similar throughout the biological world. Each ferritin is a roughly spherical, hollow protein with a thin, negatively charged shell that can hold up to 4500 Iron atoms. This shell is composed of 24 nearly Identical subunits. Junctions between three or four subunits form channels, 3-fold or 4-fold respectively, that traverse the protein shell. The hydrophilic 3-fold channels are believed to be the key pathway for the uptake and release of iron.
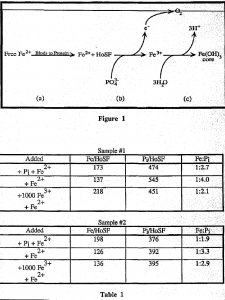
Free Iron exists as Fe” and has the potential to catalyze the formation of oxygen radicals (Fe” + 02 -> Fe3′ + 02.). Ferritin controls this toxic effect by oxidizing Iron to Fe3+ and then complexing It with water to form a stable and harmless ferric hydroxide mineral, Fe(OH) 3, inside the hollow protein. Phosphate, which is present In all native ferrltins characterized to date, plays an essential but unknown role In this process. The general pathway for Iron deposition in ferritin is shown In figure I. These steps are believed to represent the overall mechanism of Iron deposition, but the actual location of each step is not known.
Recently In our lab, we made a significant discovery concerning the role of phosphate In ferritin. Without an external oxidizing agent present to pick up the electron released, Fe2′ will not be deposited on the ferritin Interior unless phosphate Is present in the core. Upon further investigation using Moss bauer spectroscopy, we observed that the Fe2′ added was quantitatively converted to Fe”. This suggests that the added Fe” is oxidized even though no oxidizing agent Is present as an electron acceptor. We hypothesized that phosphate functions in this redox process. We focused on step (b) in the figure above.
Experiment
I used Horse Spleen Ferritin (HoSF) as a model in the proposed research and focused on determining if the phosphate remains associated with the surface of the Iron core or Is It burled upon the aerobic deposition of additional iron.
Native ferritin, which has phosphate on Its Interior (presumably bound to the Iron core surface), will bind Fe” atoms added anaerobically, while a reconstituted core without phosphate does not bind Fe” anaerobically. To answer the question, 1000 Fe” atoms per ferritin were added aerobically to native ferritin that had a phosphate layer in an attempt to bury the phosphate. The ferritin sample was then made anaerobic by pulling a vacuum on the sample to remove all oxygen present. Then excess Fe2′ was added anaerobically. There were a total of three types of ferritin for each sample: (I) excess phosphate and Fe2′ added to native HOSF, (2) only excess Fe2′ was added to the native phosphate sample, and (3) only excess Fe 2′ added to native HoSF with 1000 Fe3′ added aerobically.
Results and Discussion
The data In table 1 suggest a few conclusions. The phosphate must be on the surface of the growing core, even In the sample which had 1000 Fe3′ added aerobically. It can also be seen that little of the phosphate is lost as seen from table 1. The Fe:Phosphate ratio remains constant in all three cases studied.
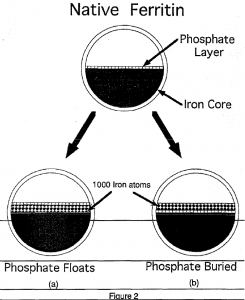
Further conclusions can be drawn from the previous conclusions based upon the data. As seen in figure 2, there are two possibilities when 1000 iron atoms are added. The phosphate can either be buried or “float” upon the core surface. The data suggest that path (b) in figure 2 is correct and that the phosphate is floating because there are binding sites only present when phosphate is present. The fact that the phosphate can float suggests that the phosphate is not strongly bound to the incoming iron. If it were, it would form a FeP04 or Fe 2(P04) 3 complex, This does not appear to be the case.
This result sheds light upon the mechanism outlined in figure 1 (b). It appears that the phosphate can interact with the iron in such a way as to allow the incoming iron to be deposited underneath and additional iron to interact with the phosphate on the core surface. This is an interesting result. Further studies will help to determine how the phosphate interacts in the redox reactions necessary for iron deposition in ferritin.
References
- Ai, M .. Muneyama, E., Kunishige, A .. and Ohdan, K. Effects of Valence States of Iron in Iron Phosphates on the Catalytic Activity in Oxidative Dehydrogenation of Isobutyric Acid. Cat. Lett. 1994, 24, 355-362.
- Ai, M., Muneyarna, E., Kunishige, A .. and Ohdan, K. Characteristics of Iron Phosphate as a Catalyst for Partial Oxidation. Bull. Chern. _Soc. _.!Jm., 1994,67, 551-556.
- Andrews, S.C., Arosio, P .. Bottke, W., Briat, J-F, von Dar!. M., Harrison, P.M., Laulhere, Levi, S., Labreaux, S., and Yewdall, S.J. Structure, Function, and Evolution of Ferri tins. I. Inorg. Biochem. 1992, 4 7, 161-17 4.
- Cheng, Y.G. and Chasteen, N.D. Role of Phosphate in Initial Iron Deposition in Apoferritin. Biochemistry 1991, 30, 2947-2953.
- Harrison, P.M., Andrews, S.C .. Artymiuk, PJ.. Ford, G. C., Guest, J.R .. Hirzmann, ]., Lawson, D.M., Livingstone, ].C., Smith, ].M.A., Treffry, A., and Yewdall, SJ. Probing Structure-Function Relations in Ferritin and Bacterioferritin. Adv. !nom. Chern. 1991,36,449-486.
- Harrison, P.M., Bauminger, E.R .. Heche!. D., Hodson, N.W., Nowik, I., Treffry, A., and Yewdall, S,j. Mechanism of FE(!!) Oxidation and Core Formation in Ferritin. Ad. Exp. MedBiol. 1994,356,1-12.
- Hequing, H., Watt, R.K., Frankel, R.B., and Watt, G.D. Role of Phosphate in Fe’• Binding to Horse Spleen Holoferritin. Biochemistry 1993,32, 1681- 1687.
- Hilty, S., Webb, B., Frankel, R.B., and Watt, G.D. Iron Core Formation in Horse Spleen Ferritin: Magnetic Susceptibility, pH, and Compositional Studies. I. Inorg. Biochem. 1994,56,173-185.
- Jemil, S., Fatemi, A., Kadir, F.H.A., Williamson, D.]., and Moore, G.R. The Uptake, Storage, and Mobilization of Iron and Aluminum in Biology. Adv. !nom. Chern. 1991,36,409-448.
- LeBrun, N.E., Cheesman, M.R .. Thomson, AJ., Moore, G.R .. Andrews, S.C .. Guest, ].R., and Harrison, P.M. An EPR Investigation of Non-haem Iron Sites in Escherichia coliBacterioferritin and Their Interaction with Phosphate: A Study Using Nitric Oxide as a Spin Probe. FEBS Leu. 1993, 323, 261-266.
- Mustafa, S, Khan, A.N., and Rehana, N. Cation-exchange Properties of Iron (III) Phosphate. I. Chern. Soc .. Faraday Trans. 1993, 89, 3843-3848.
- Watt, G.D .. Frankel, R.B .. jacobs, D. Huang, H., and Papaefthyrniou, G.C. Fe’• and Phosphate Interactions in Bacterial Ferritin from Azotobacter vinelandii. Biochemistry 1992, 31, 5672-5679.
- Watt, R.K .. Frankel, R.B., and Watt. G.D. Redox Reactions of Apo Mammalian Ferritin. Biochemistry 1992, 31, 9673-9679.