Christina Johnson and Dr. Steve Fleming, Department of Chemistry and Biochemistry
Introduction
Photoaffinity labeling is a useful methodology for determining the active site of a given enzyme. It is also a very important tool in discovering what roles different enzymes play in the human body. Our research group has been involved in a continuous effort to develop new and more effective photoaffinity labeling reagents. I A photoaffinity labeling model compound currently under investigation is the target molecule 4-nitro-3-trifluoromethylbenzyl phenyl sulfide.
Our goal is to synthesize an analogous compound, using readily available reagents, and then using those same steps, produce the target molecule. The analogous compound is pnitrobenzyl phenyl sulfide.
Experimental
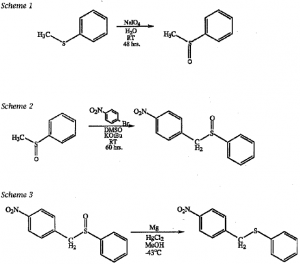
Step 1: By an oxidation reaction the sulfur of thioanisole (purchased from Aldrich) was oxidized as shown in scheme I. The oxidation was accomplished by using the reagent sodium meta-periodate, with water at room temperature, followed by stirring for forty-eight hours. The crude yield was quantitative.
Isolation of the sulfoxide included filtering the mixture and washing the filtrate with methylene chloride three times. The aqueous layer was then extracted four times with methylene chloride. The extracts were combined and the solvent was evaporated off. The sulfoxide is a light yellow oil that crystallizes upon cooling.
Step 2: To p-nitrobromobenzene in DMSO and potassium t-butoxide was added the sulfoxide. The mixture was stirred for sixty hours at room temperature. 3 This step gave a yield of 30%.
Isolation of the product involved first adding 5% hydrochloric acid to the mixture and then extracting the solution three times with ethyl ether. The solvent was then evaporated off. The product was then separated from the starting material by using column chromatography with chloroform as the solvent.
Step 3: This step will involve reduction with Mg and HgCI2 in methanol at – 43 OC.4 The isolation and purification of the resulting end product, pnitrobenzyl phenyl sulfide, will provide us with the compound analogous to the target molecule.
Summary and Future Work
Using the steps outlined in the experimental section we will be able to synthesize the analogous compound and using the same steps synthesize the target molecule. In the future step 3 and steps 1-3 using 4-nltro-3-trifluoromethylbenzyl phenyl sulfide, rather then pnitrobromobenzene, will be carried out. After the target molecule is produced in significant quantities it can be tested for its effectiveness as a photoaffinity labeling compound.
References
- Fleming, S.A,and A.W. Jensen. I. Org. Chern. 1993. 58:7135.
- Hiskey, R.G. and M.A. Harpold. I. Org. Chern. 1967. 32:3191.
- Makosza, M. and J, Winiarski. I. Org. Chern. 1980. 45:1534.
- Lee, G.H., E.B. Chou, E. Lee,and C.S. Pak. Tetrahedron Lett. 1994. 35:2195.