Scott H Fredrickson and Dr. Paul Savage, Chemistry
Introduction
Diaza-crown ethers have the ability to bind many metal cations. When certain side arms are attached, a fluorescence increase can be observed when the metal binds to the crown.1,2,3,4 Via this increase in fluorescence, presence of metal cations can be detected. For industrial purposes, these molecules may be important in the determination of toxic metal levels in waste streams. The use of different side arms effects the initial fluorescence observed. A side arm similar to 1 is not fluorescent. It is believed that transfer of the hydrogen atom on the hydroxyl group to the pyridine nitrogen quenches fluorescence. When a metal binds to the side arm, the metal interferes with proton transfer and thus allows fluorescence and detection of the metal. It is believed that the ammine on 1 will similarly quench fluorescence through single electron transfer of one of it’s electrons to the ð system.
Results
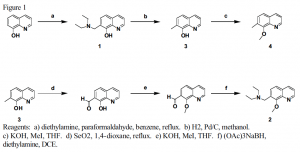
Compounds 1, 3, and 4 have been synthesized through the scheme used in figure 1. Compounds 1, 3, and 4 were characterized through 1H NMR, 13C NMR, and mass spectrometry. The synthesis of compound 2 is not completed because it may not be synthesized simply by methylating compound 1. A more complicated route of synthesis must be used.
Discussion
Once compound 3 is synthesized through the scheme in Figure 1, the fluorescence of the four compounds will be studied and compared. By comparing compounds 1 and 2 with compounds 3 and 4, the fluorescence quenching of single electron transfer will be understood. Additionally, by comparing the fluorescence of compounds 1 and 3 with compounds 2 and 4 the fluorescence quenching of proton transfer will be understood for these chemicals. Through the study of compounds 1–4, the effect of ammine and hydroxyl groups on the fluorescence of these molecules will be better understood.5
References
- Prodi, L.; Bolletta, F.; Montalti, M.; Zaccheroni, N.; Savage, P.B.; Bradshaw, J.S.; Izatt, R. M. Tetrahedron Lett. 1998, 39, 5451–5454.
- Prodi, L.; Bargossi, C.; Montalti, M.; Zaccheroni, N.; Su, N.; Bradshaw, J.S.; Izatt, R.M.; Savage, P.B. J. Am. Chem. Soc. 2000, 122, 6769–6770.
- Bronson, R.T.; Bradshaw, J.S.; Savage, P.B.; Fuangswasdi, S.; Lee, S.C.; Krakowiak, K.E.; Izatt, R.M. J. Org. Chem. 2001, 66, 4752–4758.
- Prodi, L.; Montalti, M.; Zaccheroni, N.; Bradshaw, J.S.; Izatt, R.M.; Savage, P.B. Tetrahedron Lett. 2001, 42, 2941–2944.
- Special thanks to Paul B. Savage, R. Todd Bronson, Jerald S. Bradshaw, and Reed M. Izatt.