Trevor Anderson and Professor David Busath, Department of Physiology and Developmental Biology
The influenza A virus is a severe ongoing threat to public health, accounting for over 250,000 fatalities worldwide each year. In order to actuate the virus, it is necessary for the M2 proton channel to be activated upon fusion with an endosome. This channel has been a target of antiinfluenza drugs, such as amantadine and rimantadine, to prevent such activation. However, the recent dominant point mutation, S31N, in the transmembrane domain of the proton channel have rendered amantadine and rimantadine incapable of securely binding to the region and thus ineffective at preventing the spread of the influenza A virus in a biological system. Circular dichroism spectrophotometry and fluorescence anisotropy have been used to characterize the affinity of multiple amantadine derivatives in an in vitro setting. We have identified fluorescence anisotropy as a method equally capable with circular dichroism spectrophotometry to determine the binding affinities of compounds to the M2TMD WT channel, but not the M2TMD S31N channel. The results revealed indeterminate data that suggest of the four experimental compounds tested, all of them share similar, if not stronger, affinities for the S31N as compared to rimantadine’s affinity for the M2 wild type channel.
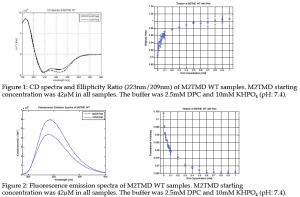
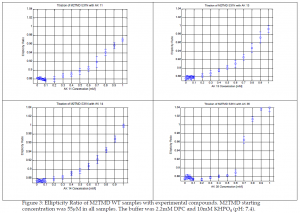
These results help to identify AK 35 as the compound most suitable to move to the next stage of animal testing and ultimately mass production due to its stronger binding affinity in comparison to the other compounds. Circular Dichroism data was applied to a titration fitting curve by converting the spectra into an ellipticity ratio, that increases as drug binds.
It is significant that while the WT FA data created a curve the appeared similar to that of the CD, the S31N didn’t create any such curve. The FA data, in conjunction with that of the emission scans, suggests a possible model for the adjustment of the tetramer upon binding to the H37xxxW41 region of the tetramer. Rather than the entire region becoming forced together, it would seem that the tetramer somewhat splays itself such that the indole group actually has a slight increased ability to move and interact with the surrounding solvent. However, this interaction is not so much of a drastic change
that would allow for a large Stokes shift to be observed in the emission spectra.
This data suggests that as drug binds to the TMD pore of the WT, that a fulcrum effect is observed that draws the TMD helix closer together but creates more room about the Trp residue, allowing for greater tumbling of the indole group. This lack of constraint on the movement of the Trp indole group, in conjunction with a negligible shift in the emission spectra (as seen in figure 2), lends to the theory that the indole group is slightly more exposed to the water within the channel, but not so much that any substantial solvent relaxation effect is observed. The S31N variant, however, observes no such fulcrum effect. As drug binds, there is no change to the amount of tumbling about the Trp indole group and is thus assumed to be have no effect, whether a tightly binding drug secures to the location of binding or not.