Peter Nielson and Dr. Scott Steffensen, Department of Psychology
Purpose
In order to further the knowledge of physiological contributions to addiction, this project’s purpose was to elucidate the neuronal processes involved in nicotine (NIC) addiction. We studied its effects on critical neuronal substrates in the ventral tegmental area (VTA), an area of the midbrain implicated in drug reinforcement. As NIC was previously shown to affect GABAergic neurotransmission, this project was to determine the more specific effect of NIC on glutamate (GLU) neurotransmission in the same area.
Procedures
Using genetically modified GAD GFP mice for identification of GABA cells, we procured 210 µm thick horizontal brain slices from around 3 week old mice, transecting the VTA. Under a microscope, UV light was used in a dark room to locate the VTA and then patch clamp into the glowing GABA cells using pipettes filled with intra-cellular fluid (ICF). Electrodes in the ICF allowed us to employ computers to measure and record changes in membrane charges and thereby measure cell firing rates. With this data and other software, counted the frequency of neuronal depolarization during time frames of control and test concentrations of NIC. Picrotoxin (PTX) was also used to isolate only the glutamate (GLU) receptors on the GABA cell bodies. The effects of different nicotinic concentrations on spontaneous excitatory post-synaptic currents (sEPSCs), or cell firing, were then measured and analyzed.
Expected Results
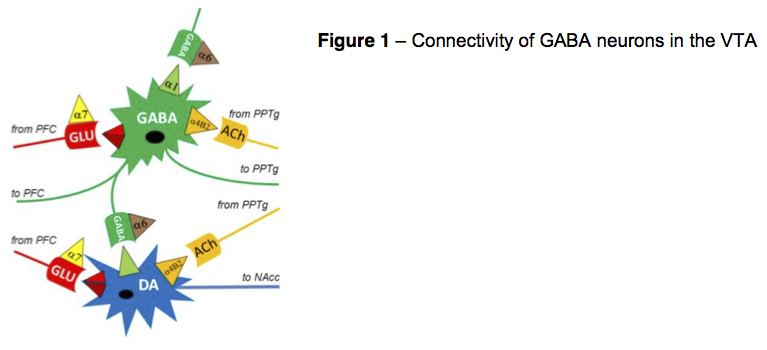
At project initiation, we expected the results of this GLU receptor isolation (red pyramid in Figure 1) to demonstrate an increase in sEPSC neuronal firing of GABA cells (green cell) at all concentrations in comparison with external control solutions.
Conclusion
The results of this study are proprietary and currently under review. They are therefore not detailed in this report.
Presentations/Publications
BYU Fulton Conference – April 2012 (partial results presented) Intermountain Premed Conference – May 2012 (partial results presented) Neuropsychopharmacology Journal – Submitted 2013
Society for Neuroscience Conference – Planned 2013
Analysis of Work Performed
We approached this project by collaboration with several student researchers to assist each other with their respective projects where similar procedures were used. We would prep solutions, maintain equipment and analyze data while efficiently procuring data from the same mouse at two workstations. We arranged our school schedules so as work simultaneously and gather more data from fewer mice.
Numerous problems presented themselves throughout the project in diverse forms. As the location for one station was shared with a researcher from another lab, oftentimes the equipment was modified or the settings were adjusted in ways that threw off measurements or caused hours of delays while we tried to determine the problems. Sometimes an overflow of extra-cellular fluid (ECF) from the cell holding tray would leave dried solutes on the lenses and mechanisms, thereby debilitating our ability to see cells clearly enough to patch clamp. Additionally, the drugs and chemicals necessary to create the experimental fluids and affect the mice were often expensive and shared with other teams working in the lab and would occasionally run out, causing an experiment to be postponed. Yet another ongoing challenge was to keep the cells alive long enough to patch, and then once recording, maintain the patch long enough to run several different solutions over the slices to obtain recordings before the seals would break. Finally, we encountered a problem in the form of conflicting results. Sometimes running two identical experiments would lead to different outcomes, causing us to analyze minute differences in mice, check drug effectiveness, change general locations in the VTA for testing, question cell sub-types in the VTA, and procure more data to solidify our answers. Consistency, patience, and perseverance were key ingredients in the overall success of this research.
We performed our final data analysis by automatically, and sometimes manually, counting the number of action potentials occurring on the measured cells under the various types of solutions. Tracking all results in a common spreadsheet, we then calculated average amplitude and frequencies of the firing until obtaining sufficient statistical data to indicate results worthy of submission to peer reviewed journals.
Future Implications
When it comes to further research, there is much yet to do in the form of isolating and analyzing the effects of NIC on other receptors and their subtypes. Testing other drugs, each in differing concentrations, and in chronic effects (ours was acute) also multiplies the amount of work to be done. As further discoveries are made in the underlying processes to addiction on the molecular level, measures may be taken to both prevent negative physiological changes and counteract ongoing influences. Drugs may potentially be developed to assist in overcoming the associated weaknesses of addiction and its effects.
Acknowledgments
Patch clamping prep work, experiments and analysis were also performed and assisted with by Brett Larsen and Jennifer Blanchard under the direction of research director Scott Steffensen. Important experimental outcomes helping support the realization and understanding of this experiment came from other groups of students and full-time employees researching their respective projects (some in vivo) at other locations within the same lab.