David Brown and Dr. Robert Davis, Department of Physics and Astronomy
In my two years of doing research at BYU, I’ve learned that the only true constant in the lab is change. Success in research seems to be determined not only by our hard work and preparation but also by our ability to be flexible and adapt our work to fit the conditions. With luck, the progress we do make will help work towards our overall goal, even if we have to use unexpected methods to get there. My research project this year was a perfect example of unexpected setbacks and insurmountable obstacles, but my lab experience in general has been truly positive in spite of these challenges.
The original project I set up with my mentor, Dr. Robert Davis, was an attempt to optimize another student’s chemical process from the previous year. This student developed a method to aggregate or “clump” nanotubes together using principles as simple as condensation and evaporation of organic solvent. The surface tension between droplets of solvent and nanotubes pulled the nanotubes closer and closer together as the droplet was evaporated, and the process could be repeated many times to give better results
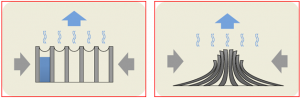
Fig 1. Diagram illustrating the aggregative effect of organic solvent condensation and evaporation on carbon nanotube forests. As the solvent evaporates, surface tension pushes the tubes closer together without dislodging them from the substrate base. (source: K. Hurd, “Process for Aggergation of Micron-sized Patterns of Vertically Alligned Carbon Nanotube Forests,” Dept. of Physics and Astronomy, Brigham Young University, August 2008.)
The proof-of-concept pictures that this student was able to take in one of BYU’s Scanning Electron Microscopes (SEM) were intriguing and inspiring. I dreamed of using this same process to strengthen carbon nanotube structures without chemically changing them, as is done in other focus groups of Dr. Davis’s nanotechnology research group. Other application ideas evolved throughout the year as we realized that this process could create tiny structural features in a way that was extremely cost-effective compared to other methods. Shrinking nanotube structures through aggregation, then, would not be simply a different way to do things on the micro-scale, but a truly better way to approach very real scientific problems. Even NASA is interested in what we’re doing.
Unfortunately, the work I performed on this project never yielded any good fruit. Though I was able to secure lab space in the underground lab, purchase necessary gas tanks, construct required tubing and valve systems, and become trained on the sophisticated SEM, the actual heater controller machine used in the experiment had been broken sometime between last year and when I received it. The internal thermocouple was broken, which meant that the box we used to control the temperature of the gas vapors thought it was already hundreds of degrees hot inside the chamber and would not allow me to heat or cool the chamber. Other cruder methods of heating were considered but were unsatisfactory for our needs.
I’m grateful to Dr. Davis, Dr. VanFleet, and Dr. Allred for their help in trying to fix this problem. I was able to present in the Spring Research Conference about our ideas and theories, but it was very discouraging for me to not have any experimental data of my own to share, especially with all the hours I spent in the lab throughout the school year. When it became clear that there wasn’t enough time to perform our experiments, even if repairs did take place, Dr. Davis reassigned me to another section of the group, where I finished out the school year working on nanotube separation procedures with a different group of students. I was able to identify several possibilities for doing nanotube fluorescence work in the mountain west and elsewhere in the country, helping solve a problem our group had been facing most of the year. Looking at the fluorescence spectrum of different types of nanotubes will help us to confidently identify them. This is very important, because different types of carbon nanotubes have extremely different electrical properties.
Research this year was sometimes discouraging, but I loved my time in the lab, even through its frustrations. My wife and I are moving to Dallas this summer where I’ll be going to medical school, and I’m excited to try research that doesn’t involve malfunctioning electronics. I am grateful to BYU for having such an outstanding undergraduate research program and to the Office of Research and Creative Activities for funding the hours I spent away from home trying to do some good for the university.
