Dr. Young Wan Ham, Department of Chemistry and Biochemistry
Evaluation of how well the academic objectives of the proposal were met
In our 2009 MEG application, we have proposed structure-based rational design of metabolite mimics based on S-adenosylmethionine (SAM) as novel antibiotics targeting SAM-I/II riboswitches and subsequent development of fluorescence assay to evaluate binding of the designed mimics to SAM-I/II riboswitches.
First, We have successfully prepared libraries of SAM mimics by attaching (1) nucleotide-intercalating agents of various binding strengths and (2) extra aminoalkyl groups on the N-terminus of SAM.
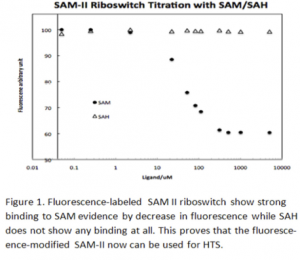
Second, a fluorescence-labeled SAM-II riboswitch was successfully prepared by replacing A9 by 2-AP, a fluorescent analogue of adenine. It was then titrated with SAM and SAH and confirmed that the fluorescence-modified SAM-II can be used for high- throughput screening (HTS) of metabolite mimics (Figure 1).
Third, the SAM mimics prepared by the structure-based rational design were screened for binding to SAM-II by fluorescence binding assay. This experiment demonstrated that interaction between SAM-riboswitches and its metabolite SAM is so delicate which is overwhelmed by intercalating-based modification leading to nonspecific binding interaction evidenced by an unexpected fluorescence increase. However, when electrostatic interaction-based mimics have shown the desired decrease in fluorescence.
Evaluation of the mentoring environment
First, the participating students, both graduates and undergraduates, involved in organic synthesis, molecular modeling, and biophysical assays. Therefore, they had ample opportunities to utilize the knowledge they had obtained in broad areas of chemistry: general, organic and analytical chemistries, and biochemistry.
As a newly starting assistant professor, it is obligatory that I, as the PI, closely work with individual students and provide necessary training and resources for the research. The PI devoted most of the midweek days in the lab to do the bench work alongside with the students. Therefore, the participating students have almost unlimited access to the PI and receive very close personal attention on their research progress. It was in addition to the individual research progress meeting held weekly.
List of students who participated and what academic deliverables they have produced or it is anticipated they will produce
List of students who participated:
Undergraduate: Susie Choi, John Mahler, Daniel Dayton, Jorden Marble, Brandon Leavitt; Graduate students: Venkatareddy Udumula, Darin Humphreys
Academic deliverables: The support from MEG resulted in 3 publications (2 published and one pending) for the participating students. One more manuscripts will be submitted in the coming months as a result of this research.
- Chittapragada, M.; Roberts, S.; Ham, Y.-W. Perspec. Med. Chem. 2009, 21-37
- Roberts, S.; Chittapragada, M.; Leavitt, B. L.; Mahler, J. W.; Ham, Y.-W.
Tetrahedron Lett. 2010, 1779-1781. - Udumula, V.; Dayton, D.; Marble, J.; Ham, Y. –W. Submitted to J. Am. Chem.
Soc.
Description of the results/findings of the project
The results mentioned in section 1 serve as a proof-of-concept in applying rational approach in designing RNA binding small molecules to target RNA. This implies that it is necessary to target RNA that is more rigid and therefore has more stable secondary structures. From this end, we have designed 2-deoxystreptamine (2-DOS) mimics to develop sequence-specific RNA binding small molecules using amide backbone to increase backbone rigidity therefore increase binding specificity. We have designed dimeric 2-DOS mimic with amide bond in between. It is shown that this mimic recognize 5’-GUUU-3’ sequence selectively. To the best knowledge of the PI, this is the first example showing that four consecutive RNA nucleotides may be targeted sequence- selectively and amide bond can be utilized to promote the selectivity. This result opens new avenue to reinvent aminoglycoside antibiotics potentially to combat resistance. In addition, side effect such as hearing loss resulted from non-specific binding may be minimized employing this approach.
A summary of how funds were used
As initially proposed, the fund was mostly used to support two graduate students (Darin Humphreys and Venkatareddy Udumula) and two undergraduate students (John Mahler and Daniel Dayton) for the summer months of 2009 and 2010. The rest of the fund was used to purchase consumables including chemicals, solvents, and RNAs.