Henrichsen, Drew
Use of Platinum Nano Particles Synthesized on Ferritin
Subtitle (if applicable)
Faculty Mentor: John Colton, Physics
I began my project considering that since I already had experience synthesizing gold
nanoparticles on ferritin, that it would be a simple step to synthesize platinum
nanoparticles. Though the materials are different, the process used by David Petrucci, a
former Ph’D student here at BYU, to synthesize platinum nanoparticles on ferritin has a
lot of similarities to what my mentor and I had done with gold. Unfortunately, though I
spoke with David Petrucci, now faculty at UVU, at length about his synthesis procedure,
I was never able to exactly duplicate his results, with both of us at a loss as to why.
Therefore, much of what I have done was discovering a number of ways that work to
accomplish the synthesis, along with finding ways to test how good a catalyst the
ferritin-platinum is.
Ferritin is a protein that looks like a hollow sphere. It is about 12 nanometers across,
and has a hole in the middle with a diameter of 8 nanometers. This hole is normally
used for holding iron in human and other mammalian blood systems. In our experiments
we tried using ferritin with and without this iron core, and as expected, found that having
the iron core correlated with greater formation of platinum nanoparticles on the surface.
The basic process used in making the nanoparticles was dissolving a form of platinum
in a buffer solution (mostly water), combining it with ferritin, and then shining a UV lamp
on it. Using 15 minutes and room temperature, like was done by David Petrucci, we
found no significant concentration of nanoparticles, so we tried many different lengths of
time and temperatures from 15 min, to 9.5 hours. After a few samples at 8+ hours, we
discovered that the ferritin broke apart after having that much exposure to either UV
light or heat, such that it was no longer useful, so we focused more on synthesizing in
the 1-6 hour range. After more analysis, I hypothesize that between 4.5 and 5 hours is
the ideal time frame for keeping the ferritin intact and allowing for the formation of
platinum nanoparticles, given the conditions of our experiment.
To study the results of the synthesis, we used an electron microscope, and the color
changes that each sample goes through when the nanoparticles form. The electron
microscope allowed us to see to sizes of a few nanometers, allowing us to see with
accuracy the quantity of platinum nanoparticles formed. Different methods of electron
microscopy also proved useful, but have no place in a paper like this.
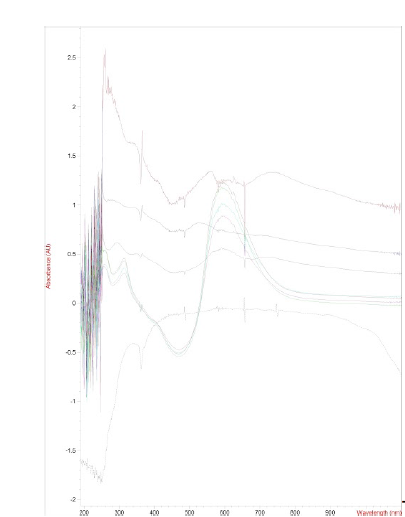
This is an example of looking at the absorbance with a
spectrometer. The absorbance is related to the color that we see when we look at an object. The
wavelength of the light increases from left to right. The colors that it absorbs are the colors we don’t see, for instance green leaves absorb red and yellow light.
A higher peak or a stronger color change indicates a greater concentration of nanoparticles. We
were able to bring the synthesis to being fairly reliable, with a good concentration of
nanoparticles, and cohesive ferritin with the help of these tools.
But, working in the lab wasn’t all serious poring over absorption charts and TEM images. On a
more humorous note, once while examining a sample underneath the electron microscope, we
saw something very much like little black sausages all over the grid that had been cut into slices
very neatly and regularly. After using diffraction techniques to figure out exactly what they were,
we found out that the dye we had been using for the entire time, had finally gotten too old, and
had formed into Uranium crystals all over the sample, then the crystals had fractured evenly as
the sample had dried. That made it look a little bit too much like this.
With the hydrogen production still in the planning states, this project is a work in progress, however, I don’t intend to leave it any time soon. Further paths that we are exploring include changing the core of the ferritin proteins, to see if the nanoparticles form better if the chemistry is different, and trying different kinds of nanoparticles.
