Alessandro Perego and John Colton, Physics and Astronomy
Introduction
Dye-sensitized solar cells (DSSCs) present a valuable and sustainable alternative to silicon solar cells. These cells present numerous advantages compered to inorganic photovoltaic systems, such as ability of absorb more sunlight per surface area than standard silicon-based solar panels, DSSCs are also able to work even in low-light conditions such as non-direct sunlight and cloudy skies. Finally, they are economical, easy to manufacture and constructed from abundant and stable resource materials. This makes DSSCs an attractive replacement for current photovoltaic technology. Ferritin (FTN) is a 12 nm diameter spherical protein with an 8 nm hollow interior, which naturally contains iron oxide nanocrystals in the form of ferrihydrite (Fe(O)OH). The natural core of ferritin can be removed and other metal oxide nanoparticles can be synthesized inside the empty ferritin. The choice of metal used in the growth of the nanoparticles determines the wavelengths of light that can be absorbed1. Recent studies also shown that the theoretical maximum efficiency of a solar device made using a combination of (Fe(O)OH), (Co(O)OH), (Mn(O)OH) and (Ti(O)OH) nanocrystals synthesized inside ferritin is up to 38% solar energy conversion6.
Additionally, ferritin possesses the ability to prevent photocorrosion in metal oxide semiconductors and it is also thermo-stable up to 80 °C. These unique properties make the ferritin nano-architecture an intriguing photocatalyst for DSSCs1.
Materials and Methods
Horse spleen ferritin (HoSF) was used as supplied for the Fe(O)OH (native nanocrystal) cores. In order to synthesize Mn, Co, and Pbs cores we followed established methods2. Anthocyanin was purified and used as a control dye for DSSC. For the anthocyanin based DSSC we soaked TiO2-coated glass plates for 30 minutes in a 0.1 mg/mL cyanin extract solution. Adsorption of cyanin to the surface of TiO2 and complexation to TiIV sites is rapid, forming a complex capable of electron injection3. For the ferritin-based dye-sensitized solar cells the different solutions were diluted down to 0.1 mg/mL using a TRIS HCl buffer at a pH of 8.5. The electrode was then soaked in the solution for 72 hours. Once ready we assembled the cell and injected a drop of the electrolyte solution between the two electrodes and proceeded to the characterization.
Results
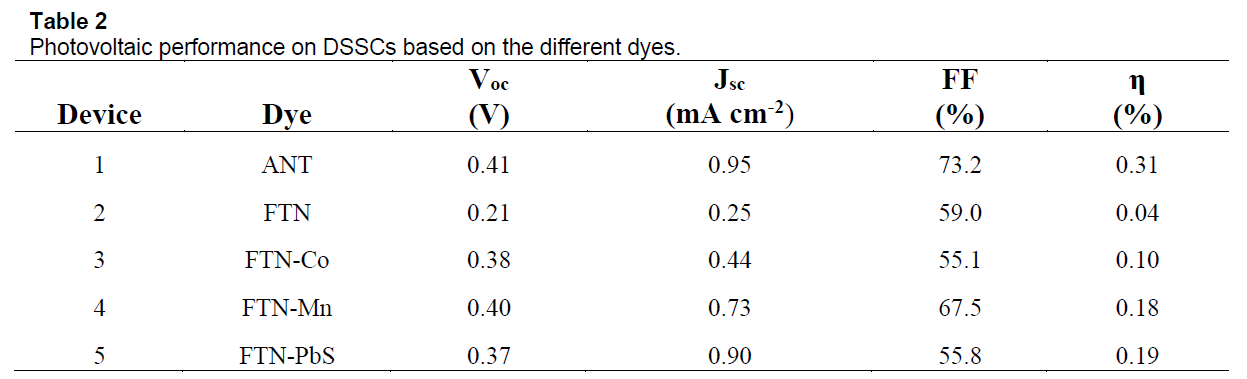
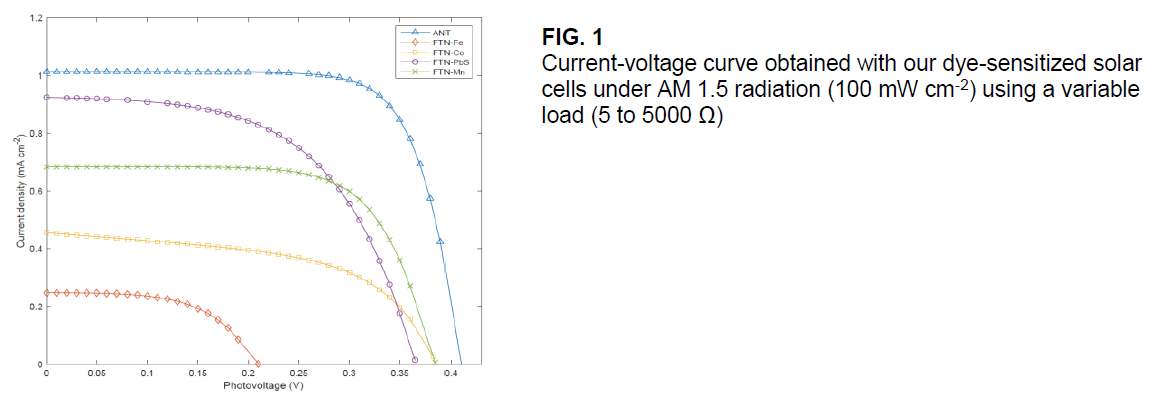
The photovoltaic properties of the solar cells constructed from these dyes were measured under exposure to the standard AM 1.5 radiation (100 mW cm-2). Figure 1 shows the I-V curve of the different cells. The measured open-circuit photovoltage (Voc), short-circuit photocurrent density (Jsc), fill factor (FF), and solar-to-electric conversion efficiencies (η) are listed in Table 1.
Conclusion
We successfully investigated the possibility of using iron, cobalt, manganese and lead-sulfide nanocrystals within ferritin as alternative dyes for dye-sensitized solar cells. Our results show that lead-sulfide and manganese nanocrystals best match the necessary properties for an alternative dye.
In order to increase the power output of the cells it is necessary to optimize and implement the deposition of ferritin on the titanium dioxide layer. Experimental analysis and computational simulation will be performed in order to better understand the physical and chemical interactions between the protein and the semiconductor material. After the optimization of a single solar cell, the next step will be to fabricate a photovoltaic module of multiple cells connected to each other. This module, called a multi-junction solar cell, will use the principle mentioned above in which different nanocrystals (namely iron, manganese, cobalt and lead-sulfide) synthesized inside ferritin can absorb different wavelengths of the light This will allowed us to harvest more frequencies of light at once and therefore, raise the efficiency of the cell.
References
- Watt, R. K., Petrucci, O. D., & Smith, T. (2013). Ferritin as a model for developing 3rd generation nano architecture organic/inorganic hybrid photo catalysts for energy conversion. Catal. Sci. Technol. Catalysis Science & Technology, 3(12), 3103. doi:10.1039/c3cy0053
- Erickson, S. D., Smith, T. J., Moses, L. M., Watt, R. K., & Colton, J. S. (2014). Non-native Co-, Mn-, and Ti-oxyhydroxide nanocrystals in ferritin for high efficiency solar energy conversion. Nanotechnology, 26(1), 015703. doi:10.1088/0957-4484/26/1/015703
- Cherepy, N. J., Smestad, G. P., Grätzel, M., & Zhang, J. Z. (1997). Ultrafast Electron Injection: Implications for a Photoelectrochemical Cell Utilizing an Anthocyanin Dye-Sensitized TiO 2 Nanocrystalline Electrode. The Journal of Physical Chemistry B J. Phys. Chem. B, 101(45), 9342-9351. doi:10.1021/jp972197w