Ryan Gillis and Dr. Alonzo Cook, Chemical Engineering
Introduction
Dry age-related macular degeneration (AMD) is a disease that causes vision loss in approximately 1.8 million Americans. Dry AMD is caused by the deterioration of the macula and associated photoreceptor cells within the retina. This degeneration occurs for a variety of reasons including aging, genetics, and retinal injury. A potential cure for dry AMD centers around the use of Müller cells, retinal cells that have the potential to transform into photoreceptor cells. If the dying photoreceptor cells can be replaced with transformed Müller cells then potentially the vision loss could be averted or reversed. With the eventual aim of a therapy using Müller cells, this work accomplished two primary goals. First, the growth rate of Müller cells was experimentally quantified in 3 environments to determine ideal culture conditions. Second, a kinetic model was created and both experimental and literature results were used to predict the potential therapeutic growth and transformation of Müller cells in the eye.
Methodology
A 25 mL cell culture flask confluent with murine Müller cells was passaged into 6 identical 25 mL flasks. Each flask was filled with one of three possible growth media (either control, nutrient rich, or nutrient poor). A microfluidic cytometer was then used to measure the cell density in each flask. Cells were allowed to proliferate and cell counts were performed regularly.
The kinetic model used for the mathematical analysis is shown below in Equations 1-5.
The model assumes that the proliferation is first order with respect to the concentrations of each cell type. [M] represents the Müller cell concentration, [MGPC] represents the concentration of Müller glial progenitor cells, while [PH] represents the concentration of photoreceptor cells. These 5 equations represent the growth, movement, and transformation of the cells. Assuming this set of differential equations as a basis, the kinetic parameter associated with Müller cell proliferation (kP) was determined by a fit of the experimental data.
Results
The first order kinetic rate constant for Müller cell proliferation was found to be 0.093 hr-1 for cells grown in 20% Fetal Bovine Serum (FBS), 0.081 hr-1 for cells grown in 40% FBS, while cells grown in solutions without FBS did not grow at all. The 20% FBS growth media was thus the most ideal solution for the mass culture of Müller cells. The kinetic constant associated with this solution implies a doubling time of approximately 7.5 hours. This is considerably faster than the 64 hour doubling time of the dedifferentiated Müller cells observed by Zhao1. The discrepancy is potentially due to the progenitor state that the cells were in that Zhao observed. Goldman observed a Müller cell doubling time of about 21 hours2. This is much closer to the value that this experiment observed. Also the discrepancies between the different literature sources suggest that a variety of factors could influence growth rates. Additionally, in the creation of a therapy these experiments would only be rough approximations of the conditions inside the eye. In this sense these experiments fulfilled their purpose by providing general estimates of the growth rate in favorable conditions.
The experimentally determined kinetic rate constant, along with approximated and literature2,3 rate constants for transformation were used to develop the theoretical cell population profile shown in Figure 1.
Discussion
The primary understanding gained from this simulation was that the most difficult step and time consuming component of this therapy will likely be the integration of the newly developed photoreceptors into the retina. The transformation and proliferation kinetics suggest that proliferation and transformation will occur much faster than integration. As such, further research should focus on working to test the ability of dedifferentiated/photoreceptor cells to integrate into an eye.
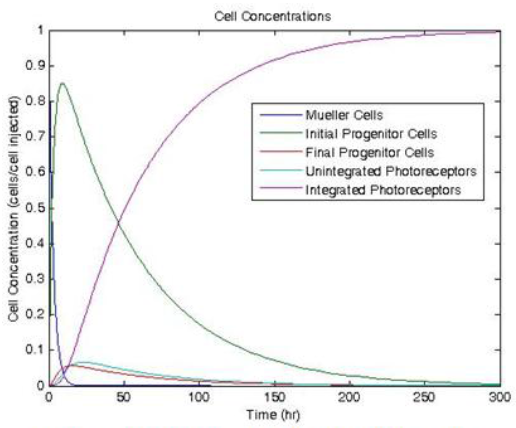
Figure 1. Modeled Cell concentration profile after injection of Müller cells
Conclusion
The final goal of Müller cell therapies for dry AMD is still somewhat distant. However, this work is a first step that both provides insight into the rates of Müller cell proliferation and predictive capability that is useful for growing Müller cells in large quantities.
References
- Zhao. JJ; et. al. Induction of Retinal Progenitors and Neurons From Mammalian Müller Glia under Defined Conditions. J Biol Chem. 289(17): 11945-51. 2014
- Goldman, Daniel; et. al. Ascl1a regulates Müller glia dedifferentiation and retina regeneration via a Lin-28-dependent, let-7 miRNA signaling pathway. Nat Cell Biol. 12(11): 1101-7. 2010.
- Limb, Astrid; et. al. Difference between the neurogenic and proliferative abilities of Müller glia with stem cell characteristics and the ciliary epithelium from the adult human eye. Exp Eye Res. 93(6):852-61. 2011.

