Jordan Eatough and Dr. Alonzo Cook, Chemical Engineering
According to the World Health Organization, cardiovascular (heart) diseases have been the number one cause of death throughout the world for over a decade. The proliferation of heart failure presents an ever-increasing demand for transplantable organs to treat such diseases. In the United States alone, more than 123,000 people are on the waiting list for a lifesaving organ transplant, with another name added every 12 minutes. Unfortunately, this demand far exceeds the available supply of transplantable organs, leading to a staggering average of 21 deaths per day1. This overwhelming deficit has prompted advancements in organ engineering as an alternative source for the ever-increasing transplant demand.
Our research team has been investigating the decellularization of porcine hearts and their subsequent recellularization with human cardiac cells. Once recellularized, we hope to use these hearts as whole-organ transplants. A major obstacle we have encountered in our pursuit to create viable organs for transplantation is the predisposition to thrombus (blood clot) formation on decellularized or improperly recellularized whole-organ specimens2. Endothelial cells, present in native tissues, are known to reduce thrombosis. Thrombosis is the formation of a blood clot inside a blood vessel, leading to the obstruction of blood flow through the circulatory system. Even with successful recellularization of a porcine heart with human cells, the organ will be useless for transplantation unless it possesses anti-thrombogenic properties.
For my ORCA, I recellularized pieces of porcine aortic extracellular matrix (ECM) with three different cell types, human umbilical vein endothelial cells (HUVECS), mouse endothelial cells (MS1), and human cardiac fibroblasts (HCF). Using fluorescent dies, I checked for successful cell growth on the ECM samples, and then applied fresh blood aliquots to each sample. I then visually qualified the amount of thrombus formation using light microscopy techniques to check for platelet adhesion to the ECM. This allowed me to compare the anti-thrombogenicity of the various recellularized ECM samples to those of both native and decellularized samples.
I developed and performed an in-vitro static blood thrombosis assay to qualitatively observe amounts of thrombosis formations. My protocol was as follows:
1. Obtain porcine aorta specimens: native aorta (positive control) and decellularized aorta (negative control).
2. Using a biopsy punch, cut small circular samples of the decellularized aorta ECM and place the samples in the wells of a 48-well plate.
3. Recellularize 14 decellularized samples each with the following cell types: HUVECS, MS1, and HCF. Label with fluorescent markers to check for cell growth (see Figure 1).
4. Fill each of the wells with an equal volume of 1 unit/ml heparinized blood.
5. Visually check for platelet adhesion after 30 minutes (see Figure 1).
6. After 1 hour, drain the blood from the wells, rinse samples, and use light microscopy techniques to more accurately check for platelet adherence to ECM. Upon visualization with light microscopy (and shown in Figure 1 below), the native ECM samples showed essentially no platelet adherence as was to be expected. The samples recellularized with HUVECS and MS1 showed slightly larger amounts of platelet adhesion as compared to the native ECM. Samples recellularized with HCF’s exhibited noticeable platelet adherence. The decellularized negative control showed the highest amount of platelet adherence.
I expected the samples recellularized with HUVECS and MS1 to be similar to native porcine aorta specimens in portraying low levels of thrombus formation. I was thus slightly surprised to see some platelet adhesion to these samples. I believe this can be contributed to the complex cellular makeup of native tissues. There are likely other factors contributing to the anti-thrombogenic properties of tissues than simply the presence of endothelial cells (HUVECS and MS1 are both endothelial cell types). Consistent with our results, I expected platelet adherence to the HCF samples as fibroblasts are an essential part of thrombus formation. I also suspected the decellularized samples to exhibit higher thrombosis levels as it contained no anti-thrombogenic properties.
In the future, I want to integrate histology and scanning electron microscopy (SEM) techniques to more precisely examine platelet adhesion at the cellular level. With time it will be possible to even more accurately qualify thrombogenicity through using fluorescent markers to precisely identify the presence of platelets. This research will be significant because thrombosis testing has not previously been performed on reendothelialized samples of full-scale porcine hearts that have potential to be used in human whole-organ transplants.
Thrombus formation, as defined by the presence of platelet adherence to the ECM and visualized using light microscopy, was found to be present in each of the samples we tested. With our results, I believe this experiment to be valid as our positive control (native ECM) showed no thrombus formation while the negative control (decellularized ECM) showed the most. The varied recellularized samples showed intermediate thrombogenic properties consistent with the current knowledge of endothelial cells reducing thrombus formation and fibroblast cells promoting thrombogenesis.
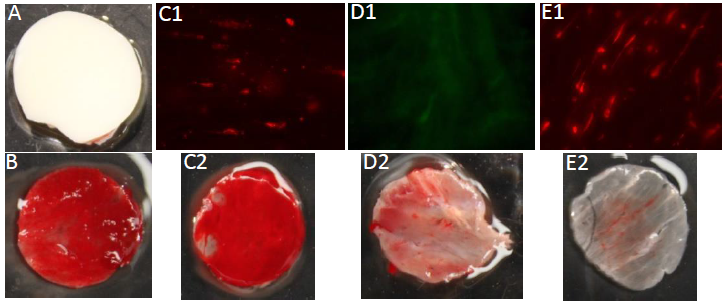
Figure 1 – Porcine aortic samples. A) Native ECM (+ control); B) Decellularized ECM thrombosis (- control); C1) Fluorescence imaging showing successful HCF proliferation on tissue; C2) HCF EMC thrombosis; D1) Fluorescence imaging showing successful proliferation of MS1 cells on tissue; D2) MS1 ECM thrombosis; E1) Fluorescence imaging showing successful proliferation of HUVECS on tissue; E2) HUVECS ECM thrombosis. A C1 D1 B C2 D2 E1 E2
References
- Facts and Myths. (2015). Retrieved August 18, 2015, from http://www.americantransplantfoundation.org/about-transplant/facts-and-myths/
- Momtahan, N., Sukavaneshvar, S., Roeder, B., & Cook, A. (2014). Strategies and Processes to Decellularize and Recellularize Hearts to Generate
Functional Organs and Reduce the Risk of Thrombosis. Tissue Engineering Part B: Reviews.
