Tyler Okelberry and Dr. Scott Steffensen, Psychology Department
Introduction
Alcohol dependency is a serious, widespread problem in the United States. The Substance Abuse and Mental Health Services Administration reported that “In 2009, the number of persons aged 12 or older needing treatment for an alcohol use problem was 19.3 million (7.7 percent of the population aged 12 or older).” 1 The following study contributed to a growing body of research about alcohol addiction and the brain. The study focused on a part of the brain called the nucleus accumbens. The release of the neurotransmitter dopamine in the nucleus accumbens has been implicated in the mental processes of reward and motivation.2 Therefore, this study aimed to explore the regulation of the release of dopamine from neuron terminals in the nucleus accumbens in order to better understand the biological basis of addiction. In order to explore the mechanisms by which dopamine release is regulated, this study recorded the effects of two chemical compounds: dehydroepiandrosterone sulfate (DHEAS) and trilostane. DHEAS is a neurosteroid that binds to and modifies the subtype-A γ-aminobutyric acid receptors (GABAARs) on 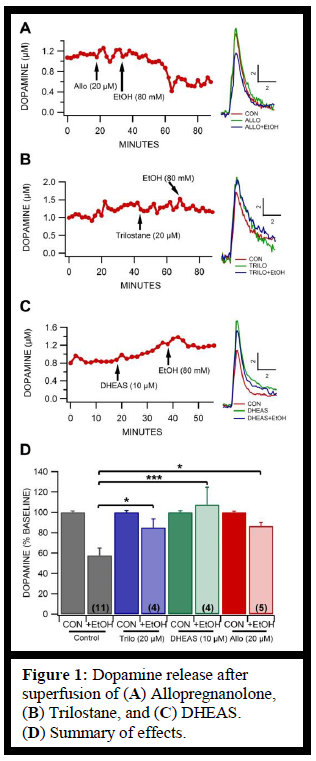 neurons.3 Trilostane is an inhibitor of an endogenous enzyme that metabolizes neurosteroids.4 Therefore, addition of Trilostane to the nucleus accumbens should increase the concentrations of endogenous neurosteroids including DHEAS.
neurons.3 Trilostane is an inhibitor of an endogenous enzyme that metabolizes neurosteroids.4 Therefore, addition of Trilostane to the nucleus accumbens should increase the concentrations of endogenous neurosteroids including DHEAS.
Methodology
This study used a technique called fast scan cyclic voltammetry (FSCV) to detect the release of dopamine from terminals in the nucleus accumbens ex-vivo. FSCV measures the amount of dopamine released in response to electrical stimulation based on current changes on a carbon fiber oxidation/reduction electrode. Data was collected by first establishing a control value for the release of dopamine. After obtaining a control value, ethanol and other compounds were superfused and the change in evoked dopamine release was recorded.
Results
DHEAS caused a moderate increase in the release of dopamine in the nucleus accumbens. In the representative recording shown in the figure (Figure 1 C), the superfusion of DHEAS caused an increase from about 0.8 μM dopamine to about 1.2 μM dopamine. In addition, DHEAS significantly attenuated the effect that ethanol normally has on dopamine release. Ethanol normally reduces the amount of dopamine released to approximately 55%, as shown in the gray control bars (Figure 1 D). Superfusion of DHEAS prior to superfusion of ethanol effectively prevented the reduction that is usually seen with ethanol. An additional neurosteroid, allopregnanolone, demonstrated a similar attenuation (Figure 1 A). Likewise, trilostane also reduced the normal effect that ethanol has on dopamine release (Figure 1 C). Whereas ethanol normally reduces the dopamine release to about 55%, ethanol preceded by trilostane reduced dopamine release to approximately 80% (Figure 1 D).
Discussion
The study’s data suggests that the regulation of dopamine in the nucleus accumbens involves, at least in part, subtype-A γ-aminobutyric acid receptors (GABAARs). These receptors are composed of a variety of subunits and it has been proposed that GABAARs containing the δ subunit are most sensitive to neurosteroids like DHEAS.5 Therefore, the results of this study suggest that GABAARs with the δ subunit play an important role in the regulation of dopamine in the nucleus accumbens.
Conclusion
Trilostane and DHEAS significantly attenuate the effect of ethanol on dopamine release in the nucleus accumbens. This observation constitutes compelling evidence that ethanol’s effect on the nucleus accumbens is linked to the δ subunits of GABAARs.
1 Substance Abuse and Mental Health Services Administration 2010. HHS Publication. SMA 10-4586 Findings. Rockville, Md, USA: Office of Applied Studies; “Results from the 2009 national survey on drug use and health: volume I. Summary of national findings.” NSDUH Series H-38A.
2 Ikemoto, Satoshi, and Jaak Panksepp. “The role of nucleus accumbens dopamine in motivated behavior: a unifying interpretation with special reference to reward-seeking.” Brain Research Reviews 31.1 (1999): 6-41.
3 Hirani, KhemrajSharma, Ajay N.Jain, Nishant S.Ugale, Rajesh R.Chopde, Chandrabhan T. 2005. “Evaluation of GABAergic neuroactive steroid 3α-hydroxy-5α-pregnane-20-one as a neurobiological substrate for the anti-anxiety effect of ethanol in rats.” Psychopharmacology 180, no. 2: 267-278. Psychology and Behavioral Sciences Collection, EBSCOhost (accessed October 24, 2013).
4 Costa, Barbara, Eleonora Da Pozzo, Beatrice Chelli, Nicola Simola, Micaela Morelli, Michele Luisi, Marco Maccheroni, Sabrina Taliani, Francesca Simorini, Federico Da Settimo, Claudia Martini, “Anxiolytic properties of a 2-phenylindolglyoxylamide TSPO ligand: Stimulation of in vitro neurosteroid production affecting GABAA receptor activity,” Psychoneuroendocrinology, Volume 36, Issue 4, May 2011, Pages 463–472.
5 Wohlfarth, Kai M., Matt T. Bianchi, and Robert L. Macdonald. “Enhanced Neurosteroid Potentiation of Ternary GABA A Receptors Containing the δ Subunit.” Journal of Neuroscience 22.5 (2002): 1541. Web.
