Gideon N. Logan and Daniel Simmons, Department of Chemistry and Biochemistry
Introduction
Cyclooxygenase (COX) isoenzymes catalyze the first step of prostanoid synthesis, which plays a role in various conditions including pain, inflammation, arthritis, and cancer. Recently our laboratory has shown that mammalian cell lines transfected with the COX-1 splice variant, COX-3, express translationally recoded proteins, including four unglycosylated lower molecular weight forms, 57kDa (57 rcCOX-3), 50kDa (50 rcCOX-3), 47kDa (47 rcCOX-3) and 44kDa (44 rcCOX-3.)
We also recently identified a synergistic effect between either 57, 50, or 44 rcCOX-3 and a cytosolic form of Nucleobindin (cNuc) to form large autophagosome structures we term mega-autophagosomes. Mega-autophagosomes formed by mouse cNuc or by mouse cNuc expressed with rat rCOX-3 proteins in Chinese Hamster Ovary (CHO) cells were found to be deficient in autophagic flux. In contrast, human forms of both rCOX-3 and cNuc expressed in CHO cells results in mega-autophagosomes where autophagic flux is not blocked.
Now that we have a grasp on the interaction of rCOX-3 and cNuc of rodent species, we need to understand why there is a functional difference in the effects of their human orthologs in evoking autophagy. Autophagy are becoming an increasingly important field of study because of their physiological roles. In fact, many viruses and bacteria replicate by hijacking cellular autophagosomes.1-3 Our laboratory’s ability to form structures similar to those used in viral/bacterial replication through the transient over-expression rcCOX-3 and cNuc demonstrates the possibility that these proteins are involved in pathogen replication. By understanding the role of the human forms of COX-3 and Nuc, we can elucidate players in replication and identify ways to block viral and bacterial spread.
Methodology
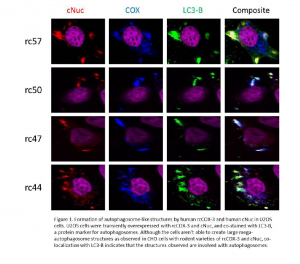
Confocal microscopy experiments were conducted to evaluate the formation and co-localization of rcCOX-3 and cNuc. In short, U2OS (human osteosarcoma) cells were plated on collagenized coverslips, transfected with rcCOX-3 and cNuc, fixed in 4% paraformaldehyde, permeabilized with Tween-20 and blocked with goat serum. The cells were incubated overnight with primary antibodies against Flag tag (rcCOX-3), Myc tag (cNuc), and LC3-B, a protein marker for autophagosomes. Cells were then stained with DAPI and respective secondary antibodies, mounted with ProLong-Gold Antifade (Invitrogen) and imaged on an Olympus FluoView 1000 confocal microscope. All experiments were performed in triplicates.
Results and Discussion
Although the U2OS cells were able to express both rcCOX-3 and cNuc, we did not observe large mega-autophagosome structures as with our previous work in CHO cells with rodent varieties of the same proteins. As seen in Figure 1, both proteins successfully co-localized with each other and also with LC3-B, but the pattern of co-localization doesn’t resemble the ring-like mega-autophagosomes. The globular masses that did form had a random distribution throughout the cell (some perinuclear, some at the periphery of the plasma membrane.) Co-localization patterns also didn’t vary between the varieties of rcCOX-3 which is consistent with our previous work.
Because rodent varieties of rcCOX-3 and cNuc were able to form large mega-autophagosomes, and human varieties of rcCOX-3 and cNuc expressed in a rodent cell line was unable to form mega-autophagosomes, we hypothesized that certain species specific chaperones were missing in order to induce the observed effect with rodent varieties. Our preliminary data using U2OS cells do not support this hypothesis.
CONFIDENTIAL PROPRIETARY INFORMATION
Conclusion
Because autophagy is a crucial part of maintaining cellular homeostasis and pathogen defense, elucidating players in the complex autophagic pathway is of utmost importance. Past work in the Simmons lab has identified both rcCOX-3 and cNuc as potent inducers of large autophagosome-like structures called mega-autophagosomes in rodent varieties. Our goal was to observe similar effects in human cell lines using human rcCOX-3 and human cNuc. Using U2OS cells, we were unable to observe induced mega-autophagosomes with human rcCOX-3 and cNuc. Further investigation with a wider range of cell lines, perhaps including primary cell lines, will help determine how mega-autophagosomes may be formed in human cells using human varieties of rcCOX-3 and cNuc.
References
1. Gutierrez, M. G. et al. Autophagy induction favours the generation and maturation of the Coxiella-replicative vacuoles. Cell Micro 7, 981-993, doi: 10.1111/j.1462-5822.2005.00527.x (2005).
2. Kemball, C.C. et al. Coxsackievirus infection induces autophagy-like vesicles and megaphagosomes in pancreatic acinar cells in vivo. J. Virol. 84, 12110-24, doi: 10.1128/JVI.01417-10 (2010).
3. Zhang, Y. et al. Autophagy promotes the replication of encephalomyocarditis virus in host cells. Autophagy 7, 613-628, doi: 10.4161/auto.7.7.15267 (2011).