Westley Kirkham and Dr. Alonzo Cook, Chemical Engineering
The removal of cells from an organ leaving behind the extracellular matrix (ECM) is termed decellularization. In the realm of tissue engineering, decellularization is important as it provides a scaffold (the ECM) onto which stem cells or differentiated cells may attach to potentially regrow an organ that has been damaged or is defunct and needs to be replaced.
The current process of decellularization, as performed in other labs, is slow and destructive to the scaffolding. With the addition of more rinse steps, different washes, and an improved bioreactor, Dr. Cook’s team has made tremendous progress in greatly decreasing the time it takes to wash a scaffold of its cells and preserving the components of the ECM. Dr. Orlando from Wake Forest University managed to decellularize a porcine kidney in 48 hours [1] of exposure to sodium dodecyl sulfate (SDS) and Dr. Sullivan at Wake Forest University was able to reduce the SDS exposure time to 36 hours [2]. Dr. Cook’s team has reduced the SDS exposure time down to 5 hours while increasing the remaining glycosaminoglycan (GAGs) content by 6 fold (see Figure 1). This was accomplished by using a feedback control on the pressure in the system (SDS only) and the use of hypertonic and hypotonic rinses in combination with SDS (tonic cycles).
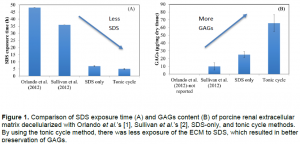
To decellularize an organ, it is attached to tubes through which washes flow. It is then set in a bath and a pump is used to pump liquid washes through the organ. Controlling the flow rate and providing an outlet matter as high pressure can rupture the organ and ruin the scaffolding provided by the matrix. Usually, what is sent through the organ is a mixture of detergent (SDS) and water. The process used in Dr. Cook’s lab includes the use of detergent in the washes but it is part of a cycle. In the tonic cycle method, saline solution and deionized water are sent through the organ alternating with SDS. This process removes cell debris as well as promoting the further lysing of cells, while preserving the GAGs, which are needed when new cells are added back to the kidney.
Previously, students had to monitor the decellularization process, manually turning valves every 15 minutes to an hour (based on the experiment we were running and the settings) until the porcine kidney had been completely decellularized and all that was left was ECM. This process could take upwards of 12 hours and in the case of doing comparative runs (how our system does vs. other’s) closer to 36 hours.
With the ORCA grant, the team was able to improve the set up in the lab for decellularizing porcine kidneys. Two hand-crafted, glass reactors with jackets for maintaining temperature in the reactor were acquired. Along with that, solid-state relays, a small heater, two small pumps for heated water to the jackets and electronic valves were added. Having two reactors allowed for more experiments. With the relays and electronic valves, the system was automated. All a lab worker had to do was turn on the pump and run the LabVIEW program and the decellularization process would proceed until someone came back to stop the run or take measurements. The electronic valves and the relays were attached to a data acquisition (DAQ) device which was attached to a laptop computer running LabVIEW software. LabVIEW was programmed so that the time of each wash could be set. The program could then control the valves through the DAQ. In this way, the experimental runs of decellularization no longer needed constant monitoring.
All of this was done with the purpose of making our decellularized organs standardized and scientifically reproducible. The bioreactor that the team had set up before the grant was an acrylic box that held a saline water bath that was open to the air. The organ was suspended in the box while it underwent decellularization. The tubing was disorganized and there was no temperature control. Valve changes had to be timed and performed manually and previous attempts at bubble traps had failed.
The new bioreactor designed and built with the aid of the grant money is much better. Having the process automated reduced human error and, with the improved reactors, reduced the number of uncontrolled variables by maintaining temperature and the pressure in the kidneys. A bubble trap was also added to keep the tubing that the washes were sent through clear of air bubbles. The bioreactors now have designated inlets and outlets for wash streams and waste streams and can be sealed so that bacteria and/or other contaminants do not get to the organ(s) being decellularized.
References
- Orlando, G., Farney AC, Iskandar SS, Mirmalek-Sani SH, Sullivan DC, Moran E, AbouShwareb T, De Coppi P, Wood KJ, Stratta RJ, Atala A, Yoo JJ, and Soker S., Production and implantation of renal extracellular matrix scaffolds from porcine kidneys as a platform for renal bioengineering investigations. Annals of Surgery, 2012. 256(2): p. 363-370.
- Sullivan, D.C., Mirmalek-Sani S-H, Deegan DB, Baptista PM, Aboushwareb T, Atala A, and Yoo JJ, Decellularization methods of porcine kidneys for whole organ engineering using a high-throughput system. Biomaterials, 2012. 33(31): p. 7756-7764.
