Cory Fronk and Professor Alonzo Cook, Chemical Engineering
Introduction
Renal failure is among the most prevalent chronic diseases in the United States today. Treatment options include blood dialysis and whole organ transplantation. Dialysis mimics the filtration function of the kidneys but cannot replicate other renal functions such as hormone secretion and homeostatic regulation. Whole organ transplantation requires dependence on immunosuppressant medication for the reminder of the recipients’ life. Also current organ supply is not sufficient to meet the needs of the individuals waiting for transplants. Bio-engineered organs offer the potential to mitigate both these issues of organ transplants. The idea is to seed a kidney shaped scaffold with renal cells from the recipient and grow a new kidney. Two forms of scaffold exist—synthetic and natural. Natural scaffolds mimic more exactly the structure and function of a normal kidney and were considered exclusively in this study. Natural scaffolds or extra cellular matrices (ECMs) support the cells in a living organ but, do not contain antigens or DNA that could cause immunologic rejection. ECM’s can only be used as scaffolds if they are completely decellularized and if they retain the peptoglycans and other proteins found on the surface. Current methods of decellularization use large quantities of ionic detergents such Sodium dodecyl sulfate (SDS) which can denature necessary proteins on the ECM and destroy the integrity of the scaffold. The purpose of this study was to determine the effectiveness of decellularization through tonic cycles.
Methodology
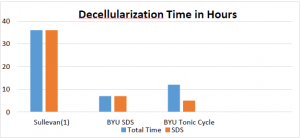
The tonic cycle method is a novel method of decellularization developed in the Tissue Engineering lab at Brigham Young University. The tonic cycle method lyses cells through changes in osmotic pressure instead of ionic detergents. In the new method, detergents are only used to clear cellular debris from the structure. The method cycles between a hypertonic solution, a hypotonic solution and an SDS solution. This sequence was found to be most effective as it introduces the greatest stress on the cell membranes. The solutions were pumped in 30 minute intervals via the renal artery. It was found that optimal decellularization occurred if the pump pressure was maintained at a constant 80 mmHg. As a benchmark, experiments using SDS without the tonic cycle were also performed. Each procedure was repeated 10 times. The average results are displayed in the figure below.
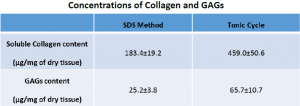
To determine the effects of SDS exposure time, each decellularized ECM was subject to several assays. Only select assays are discussed in this paper. Two assays performed were measurements of collagen and sultanate glycosaminoglycans (GAGs) concentrations in the decellularized ECM’s. The collagen assay was performed using Sircol Soluble Collagen Assay Kit (Biocolor Ltd., Newtownabbey, UK) while the GAG assay was performed with the Blyscan sGAG Assay Kit (Biocolor, Ltd). The results show significantly higher concentrations for both the collagen and the GAGs in the tonic cycle kidneys. The data is summarized below.
Results
Both methods show significant decrease in decellularization time compared to literature values. One possible explanation for this improvement is a difference in in flow rates or pressures. Another possible explanation is a deference in determination of complete decellularization. Even more interesting is the comparison between the two different methods performed at BYU. The data shows the tonic cycle to be 5 hours slower than the SDS method but, uses 2 hours less of SDS. The significance of the 2 hours can only be understood if the GAGs and collagen concentrations are considered. The 2 hour decrease caused on average a 150% increase in both the GAGS and collagen concentrations, which suggests a more fully preserved ECM.
Conclusion
The tonic cycle method proved effective at decellularization, decreased the amount of SDS used, and preserved the integrity of the ECM structure. The higher GAGs and collagen concentrations from the tonic cycle method may enable more successful recellularization. Future studies are needed to determine the actual effects of the tonic cycle method on recellularization.
References
- Sullivan DC, Mirmalek-Sani S-H, Deegan DB, Baptista PM, Aboushwareb T, Atala A, and Yoo JJ. Decellularization methods of porcine kidneys for whole organ engineering using a high-throughput system. Biomaterials 2012; 33: 7756-7764.
