Braden Tucker and Dr. Benjamin Bikman, Physiology and Developmental Biology
Introduction:
With the unabated rise in obesity in the United States and globally (1), increasing efforts are being devoted to understand and prevent both the onset and the consequences of excess fat gain. As evidenced by health trends, efforts to curtail weight gain have proven largely ineffective, as the number of obese adults in the U.S. is projected to grow by 2.4 million annually (2). The increased prevalence of obesity would not be noteworthy if such a trend were not accompanied by a substantial economic and personal cost—medical costs are roughly $150 billion annually and obese individuals may expect a loss of 20 years of life (3). Excess weight gain is associated with increased risk of the most prominent killers in the developed world, namely cardiovascular disease, type 2 diabetes, and some cancers (4). With efforts to reduce obesity failing, an alternative approach is to target the mechanism that explains the increased risk of disease with weight gain. With little exception, this mechanism is insulin resistance—a failure of particular cells to respond to insulin.
Given previous findings that ceramide biosynthesis is upregulated with weight gain (5), the aim of this project is to determine the mechanism of ceramide-induced ROS generation. Specifically, we implicate altered mitochondrial morphology as the mediator of ceramide-induced ROS release. The long-established mechanism of ceramide-induced insulin resistance, namely Akt inhibition, has recently been challenged (6), opening the possibility of an alternative mechanism. We believe altered mitochondrial morphology and ROS generation may explain ceramideinduced insulin resistance (Fig 1). Positive findings may reveal an alternative therapy to maintain/improve insulin sensitivity
Mitochondrial fission is necessary for ceramide-induced ROS generation and insulin resistance in muscle
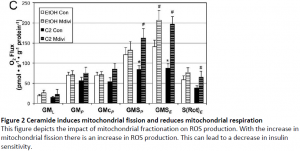
Mouse muscle cells were treated with C2 ceramide or palmatate (PA) in the presence or absence of Mdivi-1 for 12 h. Following treatment, cells were washed with PBS and exposed to Amplex Red to quantify H2O2 (ROS) production. Although C2 ceramide caused an increase in fluorescence with Amplex Red treatment, this effect was reduced by inhibition of mitochondrial fission by Mdivi-1 co-treatment. We explored the effect of blocking mitochondrial fission, with its subsequent reduction in H2O2 formation, on muscle insulin signaling. Inhibition of mitochondrial fission with Mdivi-1 protected Akt/PKB phosphorylation with insulin stimulation in C2-treated myotubes (Figure 1)
ROS generation is not necessary for mitochondrial fission or altered mitochondrial respiration with PA and ceramide treatments
To address whether ROS are necessary for the mitochondria specific effects of both ceramide and PA, we replicated earlier experiments with the addition of the glutathione precursor NAC (N-acetylcysteine), an effective reducing agent known to neutralize H2O2 [22,23]. We found that C2-treated cells that were pre-treated with NAC appeared to experience a similar degree of mitochondrial fission as the C2 treatment alone (Figure 6A). Functionally, NAC pre-treatment likewise failed to prevent reduced mitochondrial respiration in ceramide- and PA treated myotubes (Figures 6B and 6C respectively).
Conclusion
The results my study show that the increase of ceramide in muscle cells by either infusing C2 ceramide or its precursor palmatate lead to an increase production of ROS. The amount of ROS produced could be reduced if mitochondrial fission was inhibited with Mdivi. Thus, mitochondria morphology is important in ROS production and subsequent insulin sensitivity. This project is a part of a larger research project that has recently been published and can be found on Pubmed.
Sources
- Finkelstein, E.A., Trogdon, J.G., Cohen, J.W., and Dietz, W. 2009. Annual medical spending attributable to obesity: payer-and service-specific estimates. Health Aff (Millwood) 28:w822-831.
- Yanovski, S.Z., and Yanovski, J.A. 2011. Obesity prevalence in the United States–up, down, or sideways? N Engl J Med 364:987-989.
- Fontaine, K.R., Redden, D.T., Wang, C., Westfall, A.O., and Allison, D.B. 2003. Years of life lost due to obesity. JAMA 289:187-193.
- Summers, S.A. 2006. Ceramides in insulin resistance and lipotoxicity. Prog Lipid Res 45:42-72.
- Bikman, B.T., Guan, Y., Shui, G., Siddique, M.M., Holland, W.L., Kim, J.Y., Fabrias, G., Wenk, M.R., and Summers, S.A. 2012. Fenretinide prevents lipid-induced insulin resistance by blocking ceramide biosynthesis. J Biol Chem 287:17426-17437.
- Hoehn, K.L., Hohnen-Behrens, C., Cederberg, A., Wu, L.E., Turner, N., Yuasa, T., Ebina, Y., and James, D.E. 2008. IRS1-independent defects define major nodes of insulin resistance. Cell Metab 7:421-433.
- Yu, T., Sheu, S.S., Robotham, J.L., and Yoon, Y. 2008. Mitochondrial fission mediates high glucose-induced cell death through elevated production of reactive oxygen species. Cardiovasc Res 79:341-351.