Ashley Muñoz (Finch) and Dr. Bradley Geary, College of Life Sciences
Introduction
Bromus tectorum is an invasive species, commonly known as cheatgrass, that has caused major problems by disrupting the native plant ecosystem in the Intermountain West through a dramatically altered fire regime that has increased in frequency from 30-100 years to almost every 5 years (Brooks, 2002). Herbicides have been used as a form of control, but they are unsuccessful as they target only the growing plant, leaving seeds to carry over and germinate the next year, and are not selective, resulting in the death of many native plants. These problems present an urgent need to find alternative methods of control for B. tectorum.
Pyrenophora semeniperda is a naturally occuring fungus that attacks and successfully kills seeds of Bromus tectorum. P. semeniperda produces secondary metabolites that have been shown to be phytotoxic to wheat seedlings in previous research, with the primary active compound provisionally identified as cytochalasin B (Evidente et al., 2002). The goal of this project was to measure the phytotoxicity, or lethality, of metabolites produced by the fungus in liquid medium vs. in solid culture on seeds. Ten strains of P. semeniperda collected from cheatgrass seeds were tested.
Methodology
Filtrates collected from liquid cultures grown in potato dextrose broth (PDB) as well as solid cultures on autoclaved wheat seeds were previously produced for each of the 10 pathogen strains. The raw filtrate was already extracted into two portions, the lipophilic extract and aqueous remainder, by a research collaborator, Josh Nicholson, who received an ORCA grant to perform this work in 2012. He also performed the extractions of the solid cultures, which I used for this project. In the summer of 2012, I successfully developed a seedling bioassay protocol for testing the phytotoxicity of these filtrates. I performed this filtrate bioassay for pathogen strains of varying virulence.
To determine phytotoxicity, Cytochalasin B standards and organic extracts at 10-3 and 10-4 molar concentrations were first dissolved in MeOH, resulting in 7 mL of solution. 2.33 mL of each extraction solution was pipetted onto filter paper in a 10 cm plastic petri dish with 12 non-dormant cheatgrass seeds. The dishes were sealed with parafilm to maintain constant water content and incubated at 20C with a 12:12 hour photoperiod. The germination date of each individual seed was recorded and its coleoptile length measured with an electronic recording caliper five days after germination. Four replicates were included for each treatment, which also included a control solution of both PDB (for filtrate bioassays) and 1% methanol (for extract bioassays). Tests of the lipophilic fractions for the solid cultures included a positive control consisting of pure cytochalasin B. Finally, we determined if there was a relationship with the amount of cytochalasin B and the level of toxicity, or the virulence, as measured in seed inoculation bioassay tests.
Results
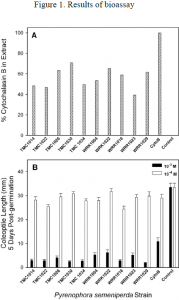
When the cytochalasin B standard and the organic extracts were tested in a seedling bioassay at 10-4 M, they were only mildly toxic; the cytochalasin B reduced coleoptile elongation to 87% of the 1% MeOH control and the ten strains of P. semeniperda were not significantly different from each other, from the cytochalasin B standard, or from the 1% MeOH control. At an equivalent concentration to 10-3 M, much higher toxicity was observed (Figure 1A). The cytochalasin B standard reduced coleoptile elongation to 34% of the control, a significant reduction. Extracts from the ten pathogen strains showed even more pronounced toxicity, reducing coleoptile elongation to 8-18% of the control. All ten extracts reduced coleoptile elongation significantly more than the cytochalasin standard, suggesting the presence of additional phytotoxic compounds. Coleoptile elongation reduction did not differ significantly among strains. There was no significant correlation between cytochalasin B concentrations in the organic extracts (Figure 1B) and their phytotoxicity in the coleoptile elongation bioassay at either 10-3 or 10-4 (Figure 1A).
Seedling bioassays of organic extracts of the filtrates produced in PDB culture also indicated high toxicity, particularly for some strains, with coleoptile elongation reduction values varying from 3 to 82% of the 1% MeOH control (data not shown). As these extracts did not contain cytochalasin B, their toxicity must have been due to the presence of other bioactive metabolites. The aqueous remainder solutions from the filtrate extractions did not exhibit any phytotoxic activity, confirming that the bioactive compounds present were lipophilic rather than hydrophilic in nature.
Discussion
There was no direct relationship between cytochalasin B concentrations in organic extracts and phytotoxicity in the seedling coleoptile elongation bioassay. Toxicity of cytochalasin B was surprisingly low compared to results reported earlier, in which a 10-4 M solution decreased root length of wheat seedlings to 62% and tomato seedlings to 28% of the control (Evidente et al.2002). Even at 10-3 M cytochalasin B had virtually no effect on B. tectorum seed germination in our experiment (i.e., seeds germinated normally), although it clearly affected 1 subsequent seedling growth. This could be because the host seeds have coverings that effectively exclude this toxin or because they have higher inherent physiological tolerance or a stronger defense response than species used in earlier bioassays, possibly because of a long shared evolutionary history with the pathogen. It is almost certain that in vivo delivery of this toxin by the pathogen inside the seed would be more effective than exogenous application.
Conclusion
This method of testing showed that we can determine the phytotoxicy, or lethality, of different metabolites produced by P. semeniperda on cheatgrass seeds. This method can be used for future metabolites that are found to be produced by this fungus, or other fungus, in determining phytotoxicity. With this knowledge, further testing can be done to determine if it is plausible to isolate these toxins and spread them directly on cheatgrass seeds to eradicate this noxious weed.