David Bilodeau and Dr. Daniel Maynes, Department of Mechanical Engineering
Background
Many metals rust when exposed to moisture and that rusting can cause the item that is made of that metal to break down. A superhydrophobic surface can repel liquids and keep metals from rusting longer. Superhydrophobic surfaces have existed in nature for a long time but are just beginning to be understood and manufactured for societal applications. They are manufactured by micro-texturing a surface that is either made from specific materials or is coated with a different material after the micro-texturing (see Figure 1). Both the material and the micro-geometry are important parameters in the surface‟s ability to repel liquids. Superhydrophobic surfaces may be used in the future as drag reducing surfaces, self-cleaning surfaces, novel heat transfer surfaces, condensers, etc. However, many of the fundamental physical laws associated with these surfaces are still largely unexplored.

When John Pearson was working on his Master‟s Thesis at BYU, he saw something new that had not been observed before. Under certain conditions when a droplet of a glycerin-water mixture struck a superhydrophobic surface, it would eject smaller droplets in a forked form (see Figure 2). The focus of this work was to quantify the existence of forked droplet rebound on the fluid properties and surface characteristics.

Experimental Procedure
Surfaces used in the experimentation were patterned with ribs. The cavity fraction which is defined as the ratio of wc/w (as seen in Figure 1) (hereafter referred to as Fc) was 0.93. The surfaces were spin coated with a 2% liquid Teflon solution and then the coating was baked on according to specifications provided by DuPont. Fluids were a combination of glycerin and water, mixed by mass (measured with 5 significant figures) which shall hereafter be denoted by the mass ratio %glycerin/% water. 0/100, 12.5/87.5, 25/75, 37.5/62.5, 45/55, 50/50, 55/45, 58.25/41.75, 67.5/32.5, 75/25, and 100/0 were tested. Viscosity (μ)* of the resulting mixture was determined by making use of the formulas presented by Nian-Sheng Cheng in his paper Formula for the Viscosity of a Glycerol-Water Mixture[1]. Surface tension was estimated using 3 known surface tensions (0/100, 50/50, and 100/0) and fitting the points with a second order polynomial. Droplet diameter was varied using a 25, 21 and 16 gauge needle and a 3mm inner diameter glass capillary.
Droplets striking the surface were recorded using a Photron FASTCAM APX RS with events recorded at 6000 fps and saved as sequential JPEG images. A MATLAB script was used to remove the background of each image and then parameters of interest (droplet location, diameter, etc.) were tracked in time with the results being saved to a Microsoft Excel file.
Results
It was our belief that the forking effect observed on the droplet splash is associated with the apparent slip** that exists on a superhydrophobic surface that is unequal along different orientations of the microscale surface patterning. We expected that the effect would be more pronounced for more viscous liquids. What we observed is that the phenomena did not appear more pronounced for more viscous liquids but that there was a range of viscosities where the phenomena occurred. Further investigation showed that the 50/50 (the only fluid John Pearson observed the phenomena occurring for) is close to the upper limit of the Oh number (a non-dimensional number that contrasts viscous forces with inertial and surface tension forces) where the phenomena is observed, but that it can be observed in much lower glycerin percentages. The results of testing variations of D (diameter) and μ (viscosity) are shown in Figure 3. The results suggested that the occurrence of the forked ejection on a ribbed superhydrophobic surface is dependent on the viscosity of the fluid, the diameter of the incoming droplet, and the surface tension between the droplet and the surrounding air.
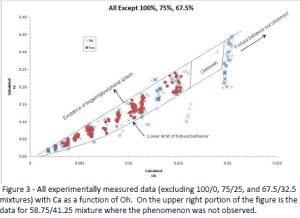
As shown in Figure 3, we also looked at the Ca number (the Capillary number represents the effect of the viscosity and the surface tension) and we then were able to isolate a range where the forking phenomena occurs. We then normalized Ca by Oh1.14 (done by Ca/Oh1.14) , with an almost constant value resulting (ranging from about 19.0 to 27.0) excluding the outlier case of the 55/45 fluid. As Oh approaches 0.02, the viscosity begins to dampen the ability of the droplet to deform into the forked shape and the phenomenon is no longer observed.
It appears that the forking is caused by the difference in droplet collapse rate. When the droplet spreads on the surface, the apparent slip is different in each direction. In one direction, there are bands of slip and no slip with the fluid moving in the direction of the bands whereas in the perpendicular direction, the fluid crosses the slip and no slip bands. Once the droplet has fully spread out, it begins to collapse as the surface tension acts to minimize the surface area of the droplet. As it is collapsing, the direction along the ribs collapses more quickly, thus driving the fluid that is collapsing across the ribs to be pushed upwards away from the surface to form the forking phenomena.
Within the range where the forked phenomenon is seen, there are still some cases where it is not seen, although the probability is quite high that it will occur in that region. Below the minimum Ca however, it was not ever observed. It appears from the data that a minimum balance of the surface tension, viscosity and velocity of the fluid exist at which point the difference in the collapse velocities in the different directions become significant and the forking phenomenon occurs. This phenomenon continues to occur until such time as the droplet begins to be unstable at the point of maximum spread (at higher incoming velocities) and the phenomena is no longer seen.
References
- Cheng, N. (2008), “Formula for the Viscosity of a Glycerol-Water Mixture”, Ind. Eng. Chem. 47, 3285-3288
